Background
Alzheimer’s disease (AD) is an irreversible, progressive brain disorder that destroys memory and cognitive function. There are currently no FDA approved disease-modifying treatments, and the reasons for the high failure rate of clinical trials in this area (99.6%: Cummings et al. 2014) are not well understood.
Incomplete understanding of disease molecular mechanisms, the timing of treatment, insufficient drug exposure to and lack of brain penetration have shown to be the contributing factor.
Results
-
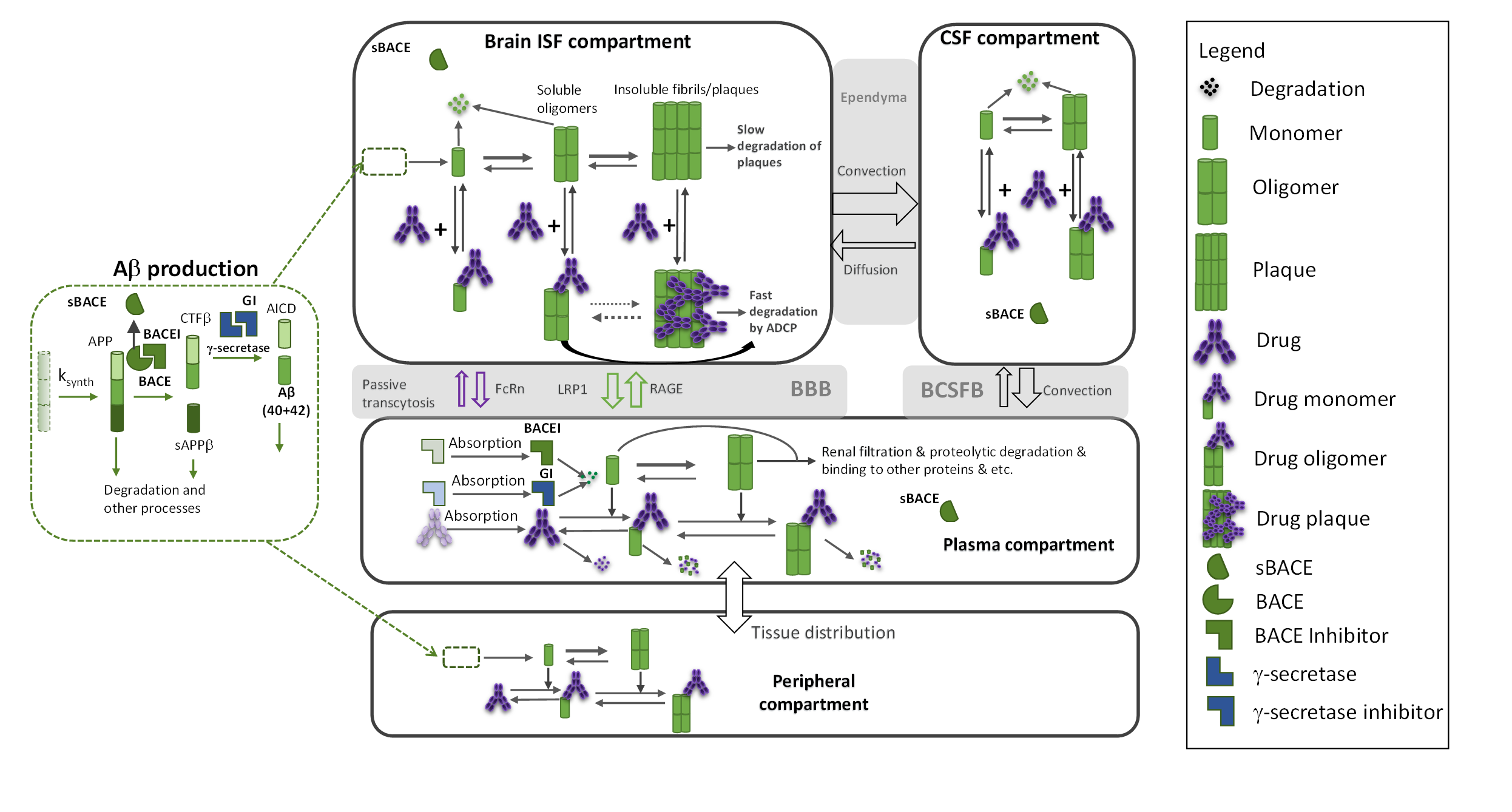
A quantitative systems pharmacology (QSP) model was developed describing the molecular mechanisms of plaque formation in AD as well as the mechanism-of-action for four anti-Aβ drugs, two b-site amyloid precursor protein cleaving enzyme (BACE) inhibitor drugs, and one g-secretase inhibitor.
-
The single QSP model captures the pharmacokinetic & pharmacodynamic data obtained with all the drugs involved in this example and is able to predict changes in different Aβ species in blood and brain, including capturing plaque reductions observed in reported clinical trials.