Applied BioMath Assess™
-
Point-and-click, model-informed drug discovery and development (MID3) software to assess efficacy, safety, and therapeutic index for early stage biotherapeutics.
-
Simulating pre-built models across a range of therapeutic modalities and disease areas helps inform target selection, project clinical dose requirements, design a lead optimization funnel, and identify areas of program risk.
Helping You Answer the Tough Questions
How hard will it be to develop the biologic (e.g., affinity, avidity, format, T1/2 dose, dose frequency, uncertainty)?
What are the pros and cons of targeting the ligand or receptor?
Should I expect Target Mediated Drug Disposition (TMDD) with this target?
Do I already have my Lead(s) or do I need to continue optimizing?
How likely is it that my dose will be too high for High Concentration Liquid Formulation?
Can my bispecific be differentiated from fixed dose combinations of monospecific antibodies?
Do I need to worry about soluble receptors for this target?
Should I expect improved affinity or PK to translate to improved dosing?
How do I think about Therapeutic Index (TI) in the design of the therapy?
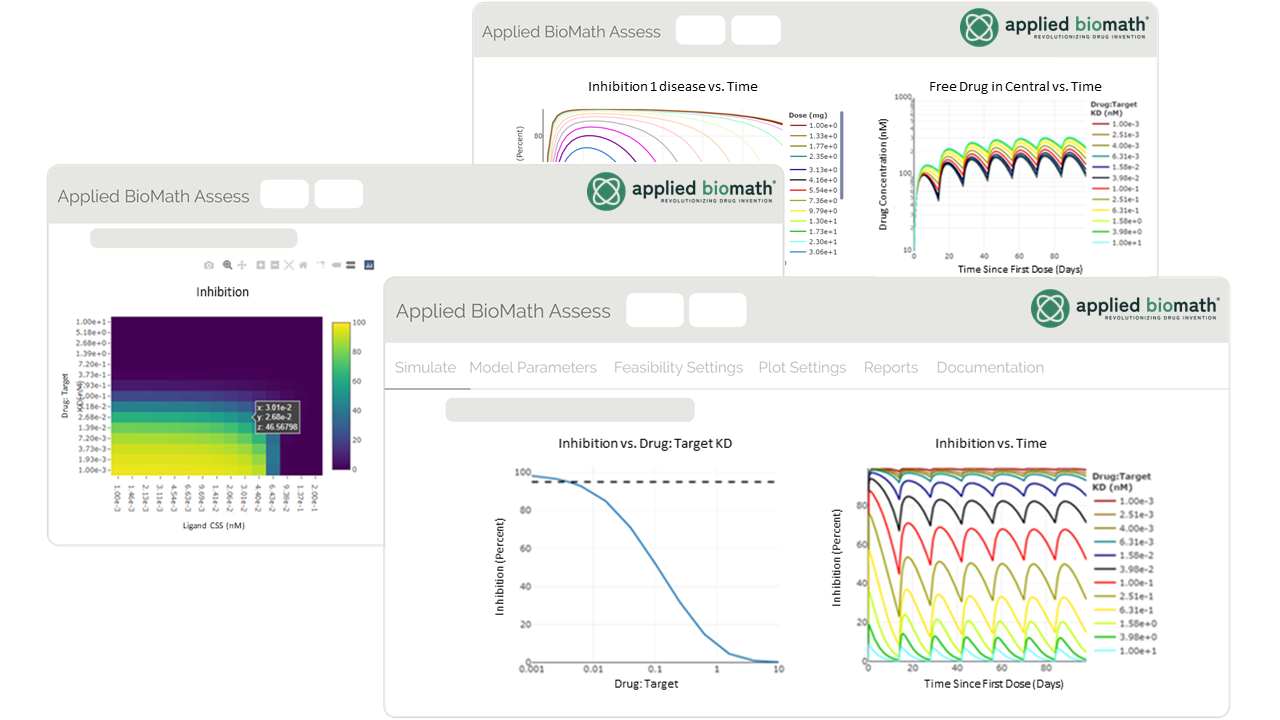
Gain Deeper Insight with Built-in Analyses and Interactive Plots
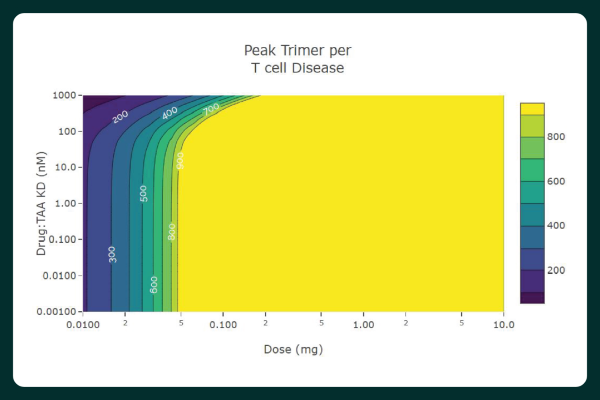
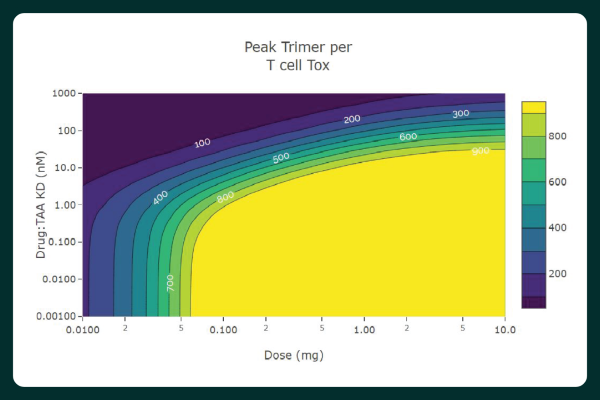
- Perform 1- or 2-dimensional scans by simply selecting scan parameters and output feasibility criteria including percent inhibition, activation, and target engagement.
- Visually assess results and key parameter values by interacting with dose-response and pharmacokinetic and pharmacodynamic (PK/PD) plots.
![]()
Get Started Quickly with Pre-built Pharmacology Models
Applied BioMath Assess™ offers several libraries of monospecific and bispecific, single compartment and multicompartment models covering common pharmacologies.
- Biotherapeutic Model Pack
- Cell Engager Model Pack
- Avidity Model Pack
- Gene Therapy Model Pack
- Protein Degrader Model Pack
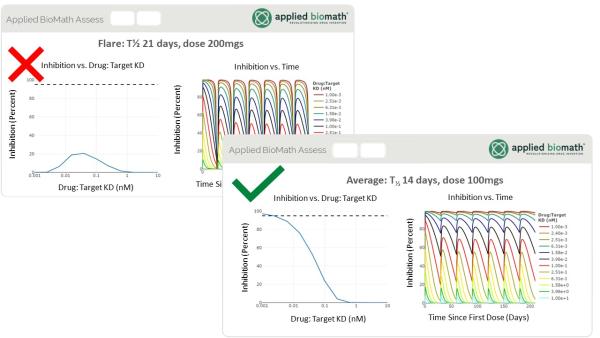
Easily Explore “What If” Questions with Scenarios
- Create Scenarios, or sets of parameters, to easily contrast and compare various scenarios.
- Add, duplicate, or delete scenarios with the click of a button in the Applied BioMath Assess interface.
- Vary parameters in the scenario table to explore risk and uncertainty while high-performance computing supports near real-time simulation when any scenario is selected.

Document, Share, and Reproduce Your Analyses
- Save scenario sets for traceability and reproducibility and/or to share work with your colleagues.
- Automatically generate a PDF report with a unique ID and date and time stamp and download plots individually as PNG images.
Biotherapeutic Model Pack
Applied BioMath Assess™ includes 18 models covering 9 common pharmacologies. Each pharmacology has a one-compartment model version and a four-compartment model version with monospecific and bispecific antibodies.
Cell Engager Model Pack
Models for solid tumors, in-vitro experiments, and solid tumors with 2 cell types.
Protein Degrader Model Pack
Includes 4 models covering in vivo and in vitro models of PROTAC and Molecular Glue Degrader pharmacologies.
Avidity Model Pack
An advanced biotherapeutic model pack that incorporates avid bivalent antibodies into many of the core biotherapeutic models.
Gene Therapy Model Pack
Models covering 8 LNP-encapsulated mRNA and siRNA therapies.
Testimonials
"Applied BioMath Assess enables me to quickly explore design hypotheses, and better understand tradeoffs between PK, Target Engagement, and TMDD of our bispecific antibodies. To be able to explore these variables in an interactive tool without having to program it from scratch is an immense time and resource saver for me. This is a powerful tool put directly in the hands of scientists."
"Applied BioMath Assess was an essential step for our portfolio prioritization. We had several therapeutics under consideration and needed to understand what parameter ranges were required to achieve our desired outcomes. Being able to quickly analyze each therapeutic helped us identify which therapeutics were best to pursue and ultimately helped us de-risk our portfolio and provide a clear and scientific rationale with our stakeholders."
Demos and Resources
Demo
Demo
Whitepaper
Support
On-Demand Support
Questions?
Looking for more information about Applied BioMath Assess?
Patent pending